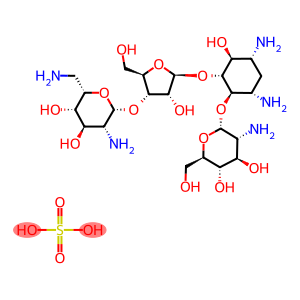
PAROMOMYCIN SULPHATE
Paromomycin sulfate
CAS: 1263-89-4
Molecular Formula: C23H47N5O18S
PAROMOMYCIN SULPHATE - Names and Identifiers
| Name | Paromomycin sulfate |
| Synonyms | fi5853 NEOMYCIN E aminosidinsulfate aminosidinesulfate aminosidinesulphate PAROMOMYCIN SULFATE Paromomycin sulfate Paromomycin Sulphate Aminosidine Sulphate PAROMOMYCIN SULPHATE paromomycin sulphates Paromomycin sulfate salt PAROMOMYCIN SULFATE SALT 4,6-diamino-2-{[3-O-(2,6-diamino-2,6-dideoxyhexopyranosyl)-D-lyxofuranosyl]oxy}-3-hydroxycyclohexyl 2-amino-2-deoxyhexopyranoside sulfate (salt) (1R,2R,3S,4R,6S)-4,6-diamino-2-{[3-O-(2,6-diamino-2,6-dideoxy-β-L-idopyranosyl)-D-ribofuranosyl]oxy}-3-hydroxycyclohexyl 2-amino-2-deoxy-α-D-glucopyranoside sulfate (salt) |
| CAS | 1263-89-4 |
| EINECS | 215-031-7 |
| InChI | InChI=1/C23H45N5O14.H2O4S/c24-2-7-13(32)15(34)10(27)21(37-7)41-19-9(4-30)39-23(17(19)36)42-20-12(31)5(25)1-6(26)18(20)40-22-11(28)16(35)14(33)8(3-29)38-22;1-5(2,3)4/h5-23,29-36H,1-4,24-28H2;(H2,1,2,3,4)/t5-,6+,7+,8-,9-,10-,11-,12+,13-,14-,15-,16-,17-,18-,19-,20-,21-,22-,23?;/m1./s1 |
| InChIKey | LJRDOKAZOAKLDU-UDXJMMFXSA-N |
PAROMOMYCIN SULPHATE - Physico-chemical Properties
| Molecular Formula | C23H47N5O18S |
| Molar Mass | 713.71 |
| Melting Point | 145 °C (decomp) |
| Boling Point | 1059.2°C at 760 mmHg |
| Specific Rotation(α) | D25 +50.5° (c = 1.5 in water pH 6) |
| Flash Point | 594.4°C |
| Water Solubility | Soluble in water |
| Solubility | Soluble in water |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | White to off-white (Solid) |
| Color | White to Off-White |
| Merck | 14,7041 |
| BRN | 5715182 |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Stability | Hygroscopic |
| MDL | MFCD00079278 |
| Use | Aminoglycoside antibiotics, can be used to treat bacterial dysentery and other gastrointestinal bacterial infections, is the treatment of amoebic dysentery |
| In vitro study | Paromycin is an aminoglycoside antibiotic with good antibacterial activity against Gram-negative and many Gram-positive bacteria, as well as some protozoa and tapeworms. In vitro analysis of the sensitivity of immastigotes in a mouse macrophage model showed that L. major and L. The tropica strain (ED50s between 1 and 5 μm) is larger than L. braziliensis (ED50, <12 μm) and L. The mexicana (ED50, 39 μm) strain was more sensitive. The strain L. donovani was moderately sensitive (ED50,6 to 18 μm), except for one Indian strain, DD8, whose ED50 was> 150 μm. |
| In vivo study | In the experimental model and CL clinical cases, the injury caused by L. major recovered faster and more completely after treatment with paromomycin ointment than the injury caused by L. amazonensis and L. panamensis. |
PAROMOMYCIN SULPHATE - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | R23/24/25 - Toxic by inhalation, in contact with skin and if swallowed. R36/38 - Irritating to eyes and skin. R39/23/24/25 - R61 - May cause harm to the unborn child R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S38 - In case of insufficient ventilation, wear suitable respiratory equipment. S36 - Wear suitable protective clothing. |
| WGK Germany | 2 |
| RTECS | WK2320000 |
| HS Code | 2941901010 |
| Toxicity | LD50 in mice (mg/kg): ~15,000 orally; 700 s.c.; 110 i.v. (Di Marco, Bertazzoli) |
PAROMOMYCIN SULPHATE - Reference
| Reference Show more | 1. [IF=7.963] Tao Wang et al."Quantitative characterization of toxicity interaction within antibiotic-heavy metal mixtures on Chlorella pyrenoidosa by a novel area-concentration ratio method."Sci Total Environ. 2021 Mar;762:144180 |
PAROMOMYCIN SULPHATE - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| Biological activity | Paromomycin Sulfate are aminoglycoside antibiotics that act on non-resistant cells and inhibit protein synthesis by binding to 16S ribosomal RNA. |
| Target | Value |
| use | amoeba protozoa must obtain metabolites of coliform flora in intestinal tract in order to survive and reproduce. Balomycin can inhibit the symbiotic bacteria of amoeba, thereby obstructing the growth and reproduction of amoeba, and indirectly exert anti-amoeba effect; in addition, it has a direct killing effect. Its strength is about twice that of emidin. It is used to treat amoebic dysentery, bacillary dysentery and enteritis. Baromomycin sulfate is an aminoglycoside antibiotic. It is a specific medicine for the treatment of amoebic dysentery. It can also be used to treat bacillary dysentery and other gastrointestinal bacterial infections. Baromomycin sulfate is an aminoglycoside antibiotic. It is a specific medicine for the treatment of amoebic dysentery, and can also be used to treat bacillary dysentery and other gastrointestinal bacterial infections. It has an inhibitory effect on amoeba and Shigella. It is mainly used for acute amoebic dysentery and bacillary dysentery; its antibacterial effect is similar to kanamycin, and is used for oral administration to treat bacillary dysentery, amoebic dysentery and enteritis. And other intestinal infections; by inhibiting the metabolism of the intestinal symbiotic flora, it indirectly exerts an anti-amoebic effect, and can also directly kill the amoebic trophoblast. |
| category | toxic substances |
| toxicity classification | low toxicity |
| acute toxicity | oral-rat LD50: 21620 mg/kg; Oral-mouse LD50: 23500 mg/kg |
| flammability hazard characteristics | combustible; combustion produces toxic nitrogen oxides and sulfur oxide smoke |
| storage and transportation characteristics | warehouse ventilation and low temperature drying |
| fire extinguishing agent | dry powder, foam, sand, carbon dioxide, mist water |
Last Update:2024-04-09 02:00:06
Supplier List
Featured ProductsMultiple SpecificationsSpot supply
Product Name: Paromomycin sulfate salt Visit Supplier Webpage Request for quotationCAS: 1263-89-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Paromomycin sulfate Request for quotation
CAS: 1263-89-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1263-89-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Paromomycin sulfate salt Visit Supplier Webpage Request for quotationCAS: 1263-89-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Neomycin Sulfate Impurity 5 Sulfate(Neomycin Sulfate EP Impurity E Sulfate)(Neomycin E Sulfate) Visit Supplier Webpage Request for quotationCAS: 1263-89-4
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Multiple SpecificationsSpot supply
Product Name: Paromomycin sulfate salt Visit Supplier Webpage Request for quotationCAS: 1263-89-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: PAROMOMYCIN SULFATE Request for quotation
CAS: 1263-89-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1263-89-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Paromomycin sulfate Request for quotation
CAS: 1263-89-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 1263-89-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Featured ProductsMultiple SpecificationsSpot supply
Product Name: Paromomycin sulfate salt Visit Supplier Webpage Request for quotationCAS: 1263-89-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Paromomycin sulfate Request for quotation
CAS: 1263-89-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1263-89-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Paromomycin sulfate salt Visit Supplier Webpage Request for quotationCAS: 1263-89-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Neomycin Sulfate Impurity 5 Sulfate(Neomycin Sulfate EP Impurity E Sulfate)(Neomycin E Sulfate) Visit Supplier Webpage Request for quotationCAS: 1263-89-4
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Multiple SpecificationsSpot supply
Product Name: Paromomycin sulfate salt Visit Supplier Webpage Request for quotationCAS: 1263-89-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: PAROMOMYCIN SULFATE Request for quotation
CAS: 1263-89-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1263-89-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Paromomycin sulfate Request for quotation
CAS: 1263-89-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 1263-89-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
View History






